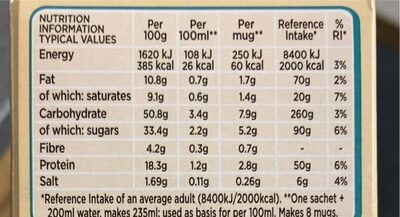
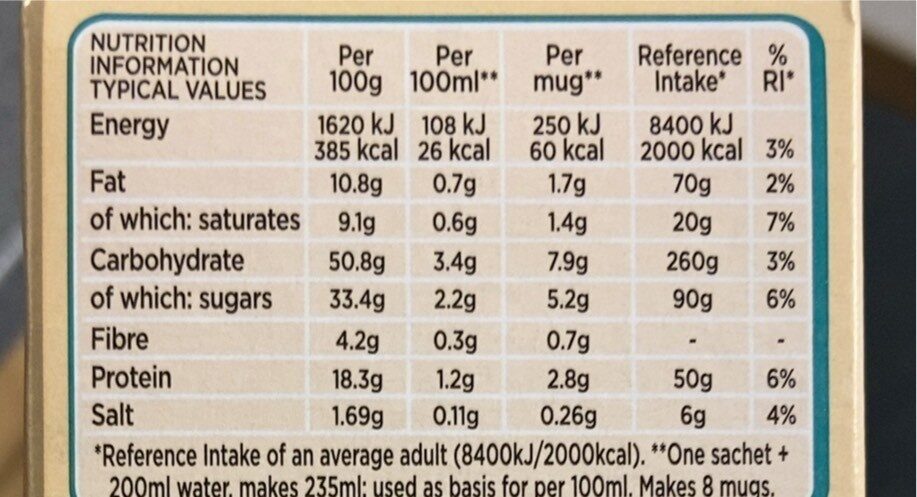
Latte - Nescafé Gold - 8 X 15.5g
This product page is not complete. You can help to complete it by editing it and adding more data from the photos we have, or by taking more photos using the app for Android or iPhone/iPad. Thank you!
×
Barcode: 7613287074638 (EAN / EAN-13)
Quantity: 8 X 15.5g
Packaging: en:Card-box, en:Unknown-packet
Brands: Nescafé Gold
Categories: en:Plant-based foods and beverages, en:Beverages, en:Plant-based foods, en:Hot beverages, en:Coffees, en:Instant beverages, en:Capsules, en:Instant coffees, en:Coffee capsules
Countries where sold: An Rìoghachd Aonaichte
Matching with your preferences
Report a problem
Data sources
Product added on by kiliweb
Last edit of product page on by sazzl3.
Product page also edited by billiebob2, inf, madeleinesmith, packbot, swipe-studio, yuka.sY2b0xO6T85zoF3NwEKvlh1Za_nbuwnkah_WhRWoyu6yF5XBZf508pfmaqo, yuka.sY2b0xO6T85zoF3NwEKvllZnctbb-gn-LDr4mHzUyem0FYPXMO5by7b5NKg.